In order for the sucrose molecules to mesh into the solution, the hydrogen bonds in the water have to be disturbed. That means that these C12H22O11 molecules end up being released into the solution.Įnergy is required to break the bonds between the molecules within sucrose, and it also requires energy to break up the oxygen-hydrogen bonds in water. When sugar is placed in the water, the bonds that hold the molecules together are easily broken up, dissolving the sugar into the water. 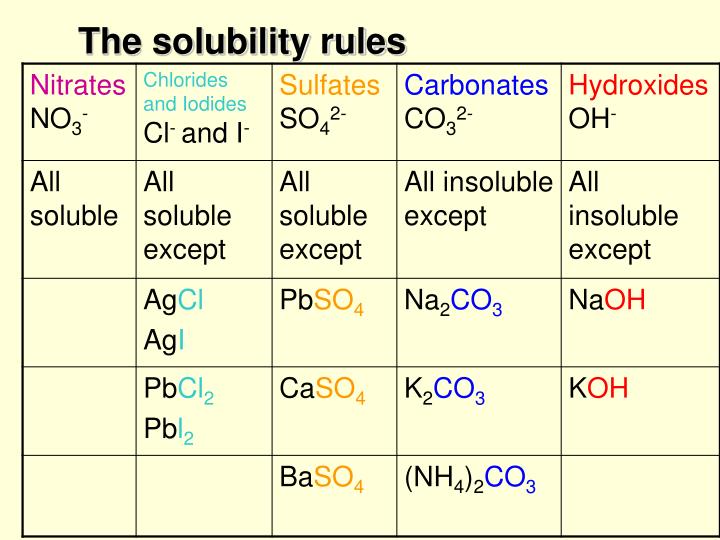
The carbon, hydrogen, and oxygen in sucrose are bonded together by inter-molecular forces, but these forces are fairly weak. Sucrose is the sugar that we use to sweeten drinks like tea and coffee. 
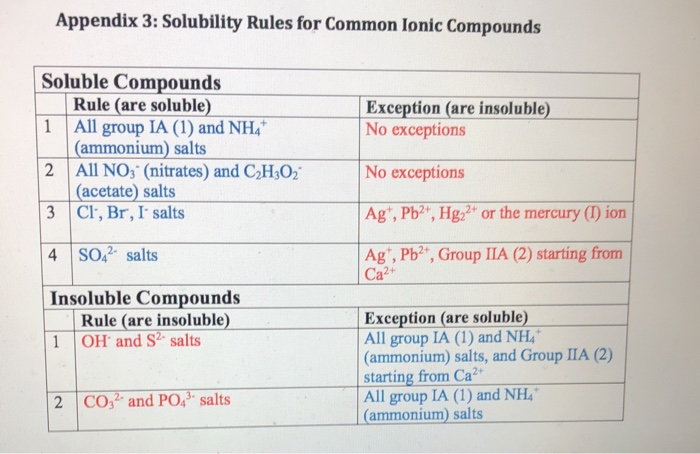
The format is X amount of solute per kilograms of solvent, or X per 100mL of solvent. In terms of measuring solubility, the solubility of a substance is often given as a concentration. Take note that this is just a heuristic, and that there are exceptions to this rule. This refers to the general rule that solvents which have similar chemical structures as the solutes they are combined with dissolve said solutes the best. When determining how likely a substance is to be soluble in another substance, the heuristic “like dissolves like” can be used. Despite the fact that the sodium chloride ions don’t dissolve like ethanol dissolves, sodium chloride is still considered water-soluble, because when the solvent evaporates crystalline sodium chloride is left as a result. These separate ions essentially just gain a coat of water molecules, becoming wrapped in them. Sodium chloride and other ionic compounds dissolve into separate ions when combined with water. In contrast, the dissolution of ionic compounds can be more complex. Covalent chemicals like ethanol create new hydrogen bonds when they dissolve in water. Dissolution is typically fairly simple when it comes to covalent chemicals. If solubility refers to substances ability to dissolve within the solute, then the actual process of the dissolving into a solvent is called dissolution. There are very few compounds which aren’t soluble at all. This term is also sometimes applied to compounds that are not easily soluble. If a chemical is not soluble, it is referred to as insoluble. The system that the solvent dissolves into, the solute, can be solid, liquid, or gas. The solvent can be either a mixture or a pure substance. Solvents are usually a solid or liquid, though not always. However, it’s an important consideration when dealing with substances like calcium sulfate which sees its solubility decreased noticeably as pressure decreases. The effect of pressure on solubility isn’t as great as temperature, and therefore it’s frequently treated as unimportant. Photo: Walkerma via Wikimedia Commons, Public DomainĪnother factor influencing the solubility of a substance is pressure. Graph depicting the relationship between temperature and solubility for various salts. In general, though, gases become more soluble when combined with organic solvents at higher temperatures, while they become less soluble in water as temperature increases. When solutes are in a gaseous form, they have hard to predict interactions with temperature increases. When liquid water is at extremely high temperatures, temperatures approaching critical temperature, ionic solutes become less soluble because of the changing properties of water at high temperatures. While most solid substances have their solubility increase as temperature increases, this isn’t universally true. The various states of matter react differently to temperature changes. As the chemical system becomes hotter, it tends to become more soluble, and as a result is capable of dissolving more solute into it then it could at lower temperatures. Meta-stability refers to a state in a chemical/dynamic system that is stable, outside of the state of least energy, or ground state.Ĭertain conditions are needed for a solution to become supersaturated, such as the solution being at the correct temperature. If the equilibrium solubility of a solution is exceeded, which can happen in certain conditions, the solution is said to be supersaturated and is metastable in nature. To be soluble in all proportions is known as miscibility. Some substances, such as ethanol and water are soluble in every possible proportion when combined with the right solvent. Photo: Vector by ZooFari raster by ZabMilenko via WIkimedia Commons, Public Domain 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
